Post Electrophoretic Analysis
Smeared Bands
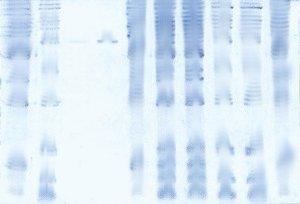
Band distortions are not staining artifacts, they occur during the electrophoretic run. Some of the most common causes are listed below:
Particulate in the sample:
Standard loading buffers are often unable to completely dissolve complex samples- fragments of bacterial cell walls, for example, can be carried over into the well. Particulate material in the well blocks proteins from entering the gel and can bind proteins, allowing them to "bleed" into the lane over time, causing characteristic streaking from the bands upward to the well. If there is particulate in the sample, a heavily stained region is often seen at the bottom of the well, where proteins were trapped, unable to enter the gel.

The 5X Protein Loading Buffer is the choice sample buffer for running your gels.
Too much sample loaded:
Overloading a well can have the same result as particulate in the sample, above. The high concentration of protein blocks the pores of the matrix, making it difficult for proteins to find a path into the gel. the result is a smearing of the bands, as proteins bleed into the gel slowly, instead of entering as a compact band. In general, limit each lane to <20ug of total protein for best results.
Poorly polymerized gel:
Polymerization of the acrylamide matrix is inhibited by oxygen. The well interfaces are particularly vulnerable to this phenomenon, as they are exposed to the plastic teeth of the comb, and/or air diffusing between the comb and the gel. If high-quality, fresh Ammonium Persulfate and TEMED are not used, the polymerization of the wells will not be complete, and the interface between the gel and the sample well will not be straight and sharp. Proteins entering such a wavy interface will form wavy bands that mimic the shape of the well. To ensure sharp banding, always use fresh APS and TEMED and allow at least 30 minutes for the gel to polymerize before running.

Protogel Sample Prep Kit concentrates and purifies your samples for best results.
Gel run too fast:
Each gel percentage, apparatus, and sample has an optimal voltage for the best electrophoretic results. Below this voltage, the run takes longer than necessary, and the bands have more time to diffuse. Above this voltage, proteins are pushed into the gel faster than they can find a viable path through the matrix. the result is that some molecules are able to move at a higher speed than others, causing smearing. Also, running at a higher voltage can cause heating, depending upon the apparatus. Uneven heating of the gel can cause local changes in buffer conductivity, leading to distorted bands.
The optimal voltage is mostly determined by the apparatus used. Check with the manufacturer to be sure your run conditions are within the recommended range for that apparatus.
 ProtoGel Quick-Cast 12%: Ready to run in 25 mins! |
 ProtoBlue Safe Colloidal Coomassie Stain |
 ProtoGel- for the best resolution SDS-PAGE |
