Post Electrophoretic Analysis Articles
Native PAGE of DNA
In the absence of denaturants double stranded DNA retains its double helical structure, which gives it a rodlike form as it migrates through a gel (for non-denaturing electrophoresis of single stranded DNA, see SSCP Analysis). Double stranded DNA of up to 1000 bp can be separated on polyacrylamide gels. DNA over 1kb is generally fractionated on agarose gels.
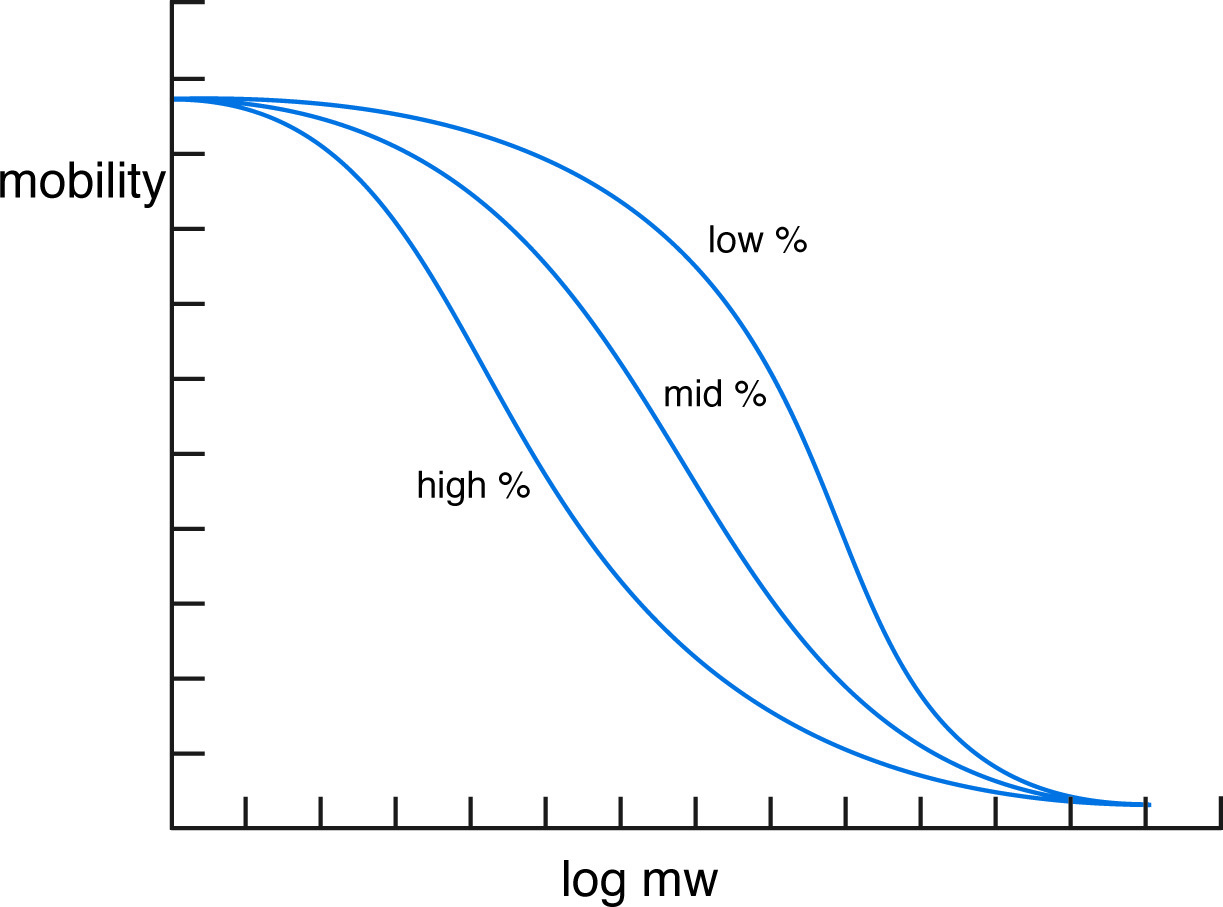
Electrophoretic mobility versus log mw.
As with single stranded DNA, double stranded DNA has a uniform negative charge density imparted by the phosphate linkages in its nucleotide backbone. As a result it has a free solution mobility (in the absence of any sieving matrix) which is independent of the size of the molecule. In a sieving medium such as polyacrylamide or agarose, the relative mobility of any given molecule is determined by its ability to find a path through the gel pores. This is a linear function of the effective radius of the molecule, which can be empirically demonstrated to be related to the logarithm of the molecular weight. Thus, on a gel of particular porosity, the migration distance of a given fragment is proportional to the logarithm of its molecular weight (log mw).
| Gel % | Size Range (bp) |
|---|---|
| 4 | 1000-2000 |
| 6 | 70-450 |
| 8 | 60-460 |
| 10 | 50-300 |
| 12 | 40-200 |
The proportionality of log mw with mobility holds within certain limits. At some small size, a molecule is no longer sieved effectively by the gel, and all molecules of that size and smaller migrate at the same rate. At some extremely large molecular size, the DNA molecule is unable to find a path through the gel, and all molecules at or above this size migrate at the same near-zero rate.
The mobility of double-stranded DNA may also be affected to some extent by the sequence of bases. Certain sequences cause bending of the helical structure, which can cause anomalously slow migration of the fragment. (Such altered mobilities serve as a basis for separation in heteroduplex analysis). Such anomalies cause problems for the determination of molecular weight with nondenaturing PAGE. The size of a fragment can be much more reliably assigned with denaturing PAGE.
DNA PAGE gels are generally run in TBE buffer, which has a high buffering capacity. TAE may also be used, but its lower buffering capacity heads to buffer exhaustion and pH changes in the gel during protracted runs. The voltages used are relatively low, around 5V/cm. With higher voltage, larger DNA molecules show a disproportionate increase in mobility, leading to a compression of bands in the higher size range.
NEXT TOPIC: Sample Prep for Native PAGE of DNA
