Oxygen Assays Articles
Immunostaining with Horseradish Peroxidase
Chromogenic and luminometric substances are also available for horseradish peroxidase (HRP). The "classic" chromogen used with HRP is diaminobenzidine (DAB). In the presence of H2O2, HRP will oxidize DAB, creating a water insoluble brown precipitate. DAB has two primary disadvantages, it is relatively insensitive and it is a potential carcinogen which must be decontaminated before disposal.
The sensitivity and convenience of HRP-based detection has been greatly enhanced by the use of luminol as a chemiluminescent substrate. HRP catalyzes the reduction of H2O2 to H2O and then recovers the electrons used for this reduction from luminol, generating an oxidized luminol radical. The luminol radical reacts with O2 to create O2-. The superoxide anion then reacts with a second luminol radical to produce a luminol endoperoxide, which subsequently decays with emission of light. The chemiluminescent reaction of HRP with luminol can be enhanced up to 104 fold by the inclusion of a variety of compounds, including substituted phenols, amines and napthalene derivatives. The mechanism of enhancement is still unclear, but it is theorized that the enhancers act as "electron conduits," channeling electrons more rapidly along the series of reactions from HRP + luminol to light emission.
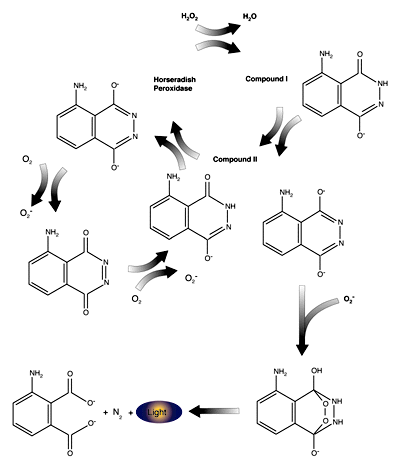
Mechanism of light emission by luminol, a luminescent substrate for use with horseradish peroxidase.
NEXT TOPIC: Enzyme Linked Immunosorbent Assay (ELISA)
